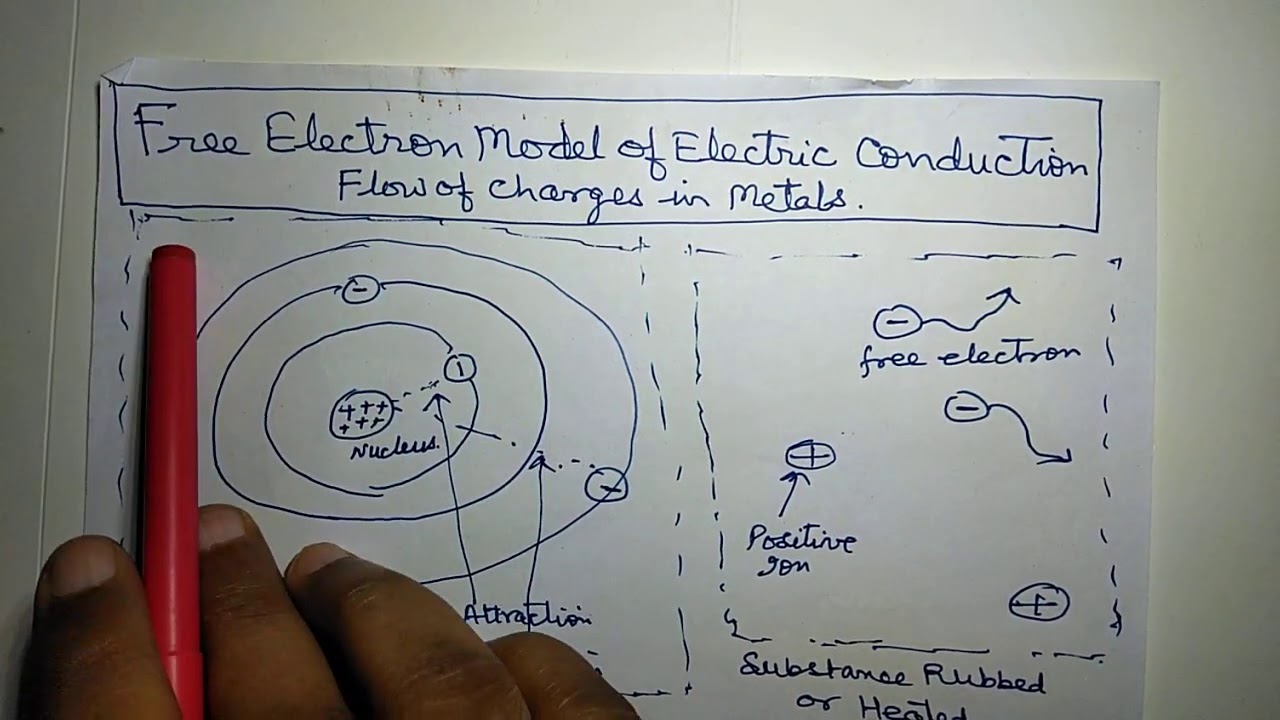
Thomson and his team of British physicists identified it as a particle in 1897 during the cathode-ray tube experiment. Irish physicist George Johnstone Stoney named this charge 'electron' in 1891, and J. In 1838, British natural philosopher Richard Laming first hypothesized the concept of an indivisible quantity of electric charge to explain the chemical properties of atoms. The exchange or sharing of the electrons between two or more atoms is the main cause of chemical bonding. Ionization or differences in the proportions of negative electrons versus positive nuclei changes the binding energy of an atomic system. The Coulomb force interaction between the positive protons within atomic nuclei and the negative electrons without allows the composition of the two known as atoms. Interactions involving electrons with other subatomic particles are of interest in fields such as chemistry and nuclear physics. Electrons are involved in many applications, such as tribology or frictional charging, electrolysis, electrochemistry, battery technologies, electronics, welding, cathode-ray tubes, photoelectricity, photovoltaic solar panels, electron microscopes, radiation therapy, lasers, gaseous ionization detectors, and particle accelerators. Special telescopes can detect electron plasma in outer space. Laboratory instruments are capable of trapping individual electrons as well as electron plasma by the use of electromagnetic fields. Electrons radiate or absorb energy in the form of photons when they are accelerated. Electromagnetic fields produced from other sources will affect the motion of an electron according to the Lorentz force law. Since an electron has charge, it has a surrounding electric field if that electron is moving relative to an observer, the observer will observe it to generate a magnetic field. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a longer de Broglie wavelength for a given energy.Įlectrons play an essential role in numerous physical phenomena, such as electricity, magnetism, chemistry, and thermal conductivity they also participate in gravitational, electromagnetic, and weak interactions. Like all elementary particles, electrons exhibit properties of both particles and waves: They can collide with other particles and can be diffracted like light. Being fermions, no two electrons can occupy the same quantum state, per the Pauli exclusion principle. Quantum mechanical properties of the electron include an intrinsic angular momentum ( spin) of a half-integer value, expressed in units of the reduced Planck constant, ħ. The electron's mass is approximately 1/1836 that of the proton. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Gaussians are typically used in molecules with three or more atoms.) is a subatomic particle with a negative one elementary electric charge. Īlthough hydrogen-like orbitals are still used as pedagogical tools, the advent of computers has made STOs preferable for atoms and diatomic molecules since combinations of STOs can replace the nodes in hydrogen-like orbitals. Each such orbital can be occupied by a maximum of two electrons, each with its own projection of spin m s. Alternative to the magnetic quantum number, the orbitals are often labeled by the associated harmonic polynomials (e.g., xy, x 2 − y 2). Įach orbital in an atom is characterized by a set of values of the three quantum numbers n, ℓ, and m l, which respectively correspond to the electron's energy, angular momentum, and an angular momentum vector component ( magnetic quantum number). The term atomic orbital may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. In atomic theory and quantum mechanics, an atomic orbital ( / ɒr b ə d l/) is a function describing the location and wave-like behavior of an electron in an atom. To see the elongated shape of ψ( x, y, z) 2 functions that show probability density more directly, see pictures of d-orbitals below. Each picture is domain coloring of a ψ( x, y, z) function which depend on the coordinates of one electron. The two colors show the phase or sign of the wave function in each region. 
The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
